

Ketoconazole 200 mg, 100 Tablets is antifungal medication used to treat yeast infections, ringworm, and other fungal infections in dogs and cats.


Learn about the prescription medication Nizoral Ketoconazole, drug uses, dosage, side effects, drug interactions, warnings, reviews and patient labeling.
KETOCONAZOLE TABLETS USP, 200 mg
Drug information on Nizoral ketoconazole, includes drug pictures, side effects, drug interactions, directions for use, symptoms of overdose, and what to avoid.
Ketoconazole Tablets should be used only when other effective antifungal therapy is not available or tolerated and the potential benefits are considered to outweigh the potential risks. Hepatotoxicity Serious hepatotoxicity, including cases with a fatal outcome or requiring liver transplantation has occurred with the use of oral ketoconazole. Some patients had no obvious risk factors for liver disease. Patients receiving this drug should be informed by the physician of the risk and should be closely monitored. See WARNINGS section. QT Prolongation and Drug Interactions Leading to QT Prolongation Coadministration of the following drugs with ketoconazole is contraindicated: dofetilide, quinidine, pimozide, cisapride, methadone, disopyramide, dronedarone, ranolazine. Ketoconazole can cause elevated plasma concentrations of these drugs and may prolong QT intervals, sometimes resulting in life-threatening ventricular dysrhythmias such as torsades de pointes. See CONTRAINDICATIONS, WARNINGS, and PRECAUTIONS: Drug Interactions sections. Ketoconazole is a weak dibasic agent and thus requires acidity for dissolution and absorption. Mean peak plasma concentrations of approximately 3.5 mcg/mL are reached within 1 to 2 hours, following oral administration of a single 200 mg dose taken with a meal. Oral bioavailability is maximal when the tablets are taken with a meal. Absorption of Ketoconazole Tablets is reduced in subjects with reduced gastric acidity, such as subjects taking medications known as acid neutralizing medicines (e.g. aluminum hydroxide) and gastric acid secretion suppressors (e.g. H2-receptor antagonists, proton pump inhibitors) or subjects with achlorhydria caused by certain diseases. (See PRECAUTIONS: Drug Interactions.) Absorption of ketoconazole under fasted conditions in these subjects is increased when Ketoconazole Tablets are administered with an acidic beverage (such as non-diet cola). After pretreatment with omeprazole, a proton pump inhibitor, the bioavailability of a single 200 mg dose of ketoconazole under fasted conditions was decreased to 17% of the bioavailability of ketoconazole administered alone. When ketoconazole was administered with non-diet cola after pretreatment with omeprazole, the bioavailability was 65% of that after administration of ketoconazole alone. Slideshow: The ABC's of Hepatitis: Get to Know This Viral Disease Distribution In vitro, the plasma protein binding is about 99% mainly to the albumin fraction. Ketoconazole is widely distributed into tissues; however, only a negligible proportion reaches the cerebrospinal fluid. Metabolism Following absorption from the gastrointestinal tract, Ketoconazole Tablets are converted into several inactive metabolites. In vitro studies have shown that CYP3A4 is the major enzyme involved in the metabolism of ketoconazole. The major identified metabolic pathways are oxidation and degradation of the imidazole and piperazine rings, by hepatic microsomal enzymes. In addition, oxidative O-dealkylation and aromatic hydroxylation does occur. Ketoconazole has not been demonstrated to induce its own metabolism. Elimination Elimination from plasma is biphasic with a half-life of 2 hours during the first 10 hours and 8 hours thereafter. Approximately 13% of the dose is excreted in the urine, of which 2 to 4% is unchanged drug. The major route of excretion is through the bile into the intestinal tract with about 57% being excreted in the feces. Special Populations Patients with Hepatic or Renal Impairment In patients with hepatic or renal impairment, the overall pharmacokinetics of ketoconazole was not significantly different when compared with healthy subjects. Pediatric Patients Limited pharmacokinetic data are available on the use of Ketoconazole Tablets in the pediatric population. Measurable ketoconazole plasma concentrations have been observed in pre-term infants (single or daily doses of 3 to 10 mg/kg) and in pediatric patients 5 months of age and older (daily doses of 3 to 13 mg/kg) when the drug was administered as a suspension, tablet or crushed tablet. Limited data suggest that absorption may be greater when the drug is administered as a suspension compared to a crushed tablet. Conditions that raise gastric pH may lower or prevent absorption (see PRECAUTIONS: Drug Interactions). Maximum plasma concentrations occurred 1 to 2 hours after dosing and were in the same general range as those seen in adults who received a 200 to 400 mg dose. Ketoconazole Tablets are indicated for the treatment of the following systemic fungal infections in patients who have failed or who are intolerant to other therapies: blastomycosis, coccidioidomycosis, histoplasmosis, chromomycosis, and paracoccidioidomycosis. Ketoconazole Tablets should not be used for fungal meningitis because it penetrates poorly into the cerebrospinal fluid. Therefore, administration of potent enzyme inducers of CYP3A4 with Ketoconazole Tablets is not recommended. The use of these drugs should be avoided from 2 weeks before and during treatment with Ketoconazole Tablets, unless the benefits outweigh the risk of potentially reduced ketoconazole efficacy. Upon coadministration, the antifungal activity should be monitored and the Ketoconazole Tablets dose increased as deemed necessary. Drug Class Contraindicted Not Recommended Use with Caution Comments Under no circumstances should the drug be coadministered with Ketoconazole Tablets, and up to one week after discontinuation of treatment with ketoconazole. The use of the drug should be avoided during and up to one week after discontinuation of treatment with Ketoconazole Tablets, unless the benefits outweigh the potentially increased risks of side effects. If coadministration cannot be avoided, clinical monitoring for signs or symptoms of increased or prolonged effects or side effects of the interacting drug is recommended, and its dosage should be reduced or interrupted as deemed necessary. When appropriate, plasma concentrations should be measured. The label of the coadministered drug should be consulted for information on dose adjustment and adverse effects. Careful monitoring is recommended when the drug is coadministered with Ketoconazole Tablets. Upon coadministration, patients should be monitored closely for signs or symptoms of increased or prolonged effects or side effects of the interacting drug, and its dosage should be reduced as deemed necessary. When appropriate, plasma concentrations should be measured. The label of the coadministered drug should be consulted for information on dose adjustment and adverse effects. Methadone: The potential increase in plasma concentrations of methadone when coadministered with Ketoconazole Tablets may increase the risk of serious cardiovascular events including QT prolongation and torsade de pointes, or respiratory or CNS depression. [See CONTRAINDICATIONS. ] Fentanyl: The potential increase in plasma concentrations of fentanyl when coadministered with Ketoconazole Tablets may increase the risk of potentially fatal respiratory depression. Sufentanil: No human pharmacokinetic data of an interaction with ketoconazole are available. In vitro data suggest that sufentanil is metabolized by CYP3A4 and so potentially increased sufentanil plasma concentrations would be expected when coadministered with Ketoconazole Tablets. eletriptan Ergot alkaloids: The potential increase in plasma concentrations of ergot alkaloids when coadministered with Ketoconazole Tablets may increase the risk of ergotism, i.e., a risk for vasospasm potentially leading to cerebral ischemia and/or ischemia of the extremities. Eletriptan: Eletriptan should be used with caution with ketoconazole, and specifically, should not be used within at least 72 hours of treatment with ketoconazole. Alprazolam, midazolam, triazolam: Coadministration of Ketoconazole Tablets with oral midazolam or triazolam, or alprazolam may cause several-fold increases in plasma concentrations of these drugs. This may potentiate and prolong hypnotic and sedative effects, especially with repeated dosing or chronic administration of these agents. Pimozide: The potential increase in plasma concentrations of pimozide when coadministered with Ketoconazole Tablets may increase the risk of serious cardiovascular events including QT prolongation and torsade de pointes. Aripiprazole: Coadministration of ketoconazole (200 mg/day for 14 days) with a 15 mg single dose of aripiprazole increased the AUC of aripiprazole and its active metabolite by 63% and 77%, respectively. The effect of a higher ketoconazole dose (400 mg/day) has not been studied. When ketoconazole is given concomitantly with aripiprazole, the aripiprazole dose should be reduced to one-half of the recommended dose. Buspirone: Ketoconazole is expected to inhibit buspirone metabolism and increase plasma concentrations of buspirone. If a patient has been titrated to a stable dosage on buspirone, a dose reduction of buspirone may be necessary to avoid adverse events attributable to buspirone or diminished anxiolytic activity. Calcium channel blockers can have a negative inotropic effect which may be additive to those of ketoconazole. The potential increase in plasma concentrations of calcium channel blockers when coadministered with Ketoconazole Tablets may increase the risk of edema and congestive heart failure. Dihydropyridines: Concomitant administration of Ketoconazole Tablets may cause several-fold increases in plasma concentrations of dihydropyridines. Ranolazine: The potential increase in plasma concentrations of ranolazine when coadministered with Ketoconazole Tablets may increase the risk of serious cardiovascular events including QT prolongation. Bosentan: Coadministration of bosentan 125 mg twice daily and ketoconazole, increased the plasma concentrations of bosentan by approximately 2 fold in normal volunteers. No dose adjustment of bosentan is necessary, but patients should be monitored for increased pharmacologic effects and adverse reactions of bosentan. Diuretics eplerenone The potential increase in plasma concentrations of eplerenone when coadministered with Ketoconazole Tablets may increase the risk of hyperkalemia and hypotension. Gastrointestinal Rapamycin (sirolimus): Ketoconazole Tablets 200 mg daily for 10 days increased the Cmax and AUC of a single 5 mg dose of sirolimus by 4.3 fold and 10.9 fold, respectively in 23 healthy subjects. Fluticasone: Coadministration of fluticasone propionate and ketoconazole is not recommended unless the potential benefit to the patient outweighs the risk of systemic corticosteroid side effects. Ketoconazole Tablets, USP What is the most important information I should know about Ketoconazole Tablets? Ketoconazole Tablets are not the only medicine available to treat fungal infections and should only be used when other medicines are not right for you. Talk to your healthcare provider to find out if Ketoconazole Tablets are right for you. Ketoconazole Tablets can cause serious side effects, including: Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using Ketoconazole Tablets with certain other medicines may affect each other. Using Ketoconazole Tablets with other medicines can cause serious side effects. How should I take Ketoconazole Tablets? The most common side effects of Ketoconazole Tablets include nausea, headache, diarrhea, stomach pain, and abnormal liver function tests. These are not all the possible side effects of Ketoconazole Tablets. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store Ketoconazole Tablets? General information about the safe and effective use of Ketoconazole Tablets. Medications are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Ketoconazole Tablets for a condition for which they were not prescribed. Do not give Ketoconazole Tablets to other people, even if they have the same symptoms that you have. They may harm them. This Medication Guide summarizes the most important information about Ketoconazole Tablets. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about Ketoconazole Tablets that is written for health professionals. What are the ingredients in Ketoconazole Tablets, USP? Active ingredient: ketoconazole. Inactive ingredients: colloidal silicon dioxide, corn starch, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and povidone. This Medication Guide has been approved by the U.S. Food and Drug Administration TEVA PHARMACEUTICALS USA Sellersville, PA 18960 Rev. B 3/2014','url':'http://www.drugs.com/pro/ketoconazole-tablets.html','og_descr':'Ketoconazole Tablets official prescribing information for healthcare professionals. Includes: indications, dosage, adverse reactions, pharmacology and more.
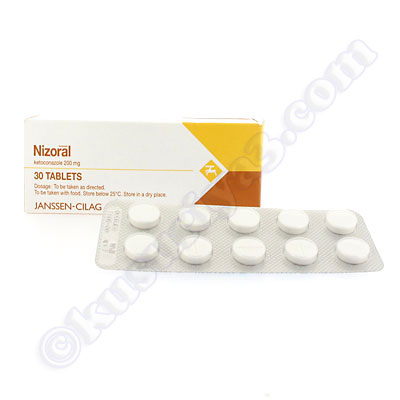
NIZORAL 200 mg tablet COMPOSITION Each tablet contains ketoconazole 200 mg. PHARMACOLOGICAL CLASSIFICATION A.20.2.2 Antimicrobial chemotherapeutic.

Ketoconazole is a synthetic broad-spectrum antifungal agent. Each tablet, for oral administration, contains 200 mg ketoconazole base. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, corn starch, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and povidone. Ketoconazole is cis-1-Acetyl-4-[p-[[2-(2,4-dichlorophenyl)-2-(imidazol-1-ylmethyl) ...
- Ketoconazole Tablets official prescribing information for healthcare professionals. Includes: indications, dosage, adverse reactions, pharmacology and more.
- DESCRIPTION. Ketoconazole is a synthetic broad-spectrum antifungal agent. Each tablet, for oral administration, contains 200 mg ketoconazole base.
- Find patient medical information for ketoconazole oral on WebMD including its uses, side effects and safety, interactions, pictures, warnings and user ratings.




















 RSS Feed
RSS Feed
